Watch batteries used in quartz watches, specifically Silver Oxide cells, often leak. (click here to learn more about watch batteries)
The electrolyte used in a Silver Oxide watch battery is highly alkaline and corrosive and so when a battery leaks inside a watch it can be catastrophic for the movement.
In a combined survey of over 3000 watches where the battery had expired, the percentage of those expended batteries that had leaked was more than 40%. (The full conclusions of the survey are towards the end of this article).
Of those leaked batteries, a third, or between 12-15% of the total number of watches examined, were sufficiently damaged to warrant either a full-service or total replacement of the watch movement.
In the remaining two thirds of the cases where the battery had leaked, the damage done was not severe enough to warrant a service of the watch or an exchange of movement, but rather a simple clean-up, which will be demonstrated later where I will show you how to clean the battery terminals in a watch.
As you can see, this under-reported problem can be the cause of great expense and inconvenience to the owner of a quartz watch. There are however certain steps you can take to limit your exposure to any potential problems, which this article hopes to introduce.

Why do Watch Batteries Leak?
To learn why Silver Oxide batteries leak so frequently, it is important to understand how the battery is assembled.
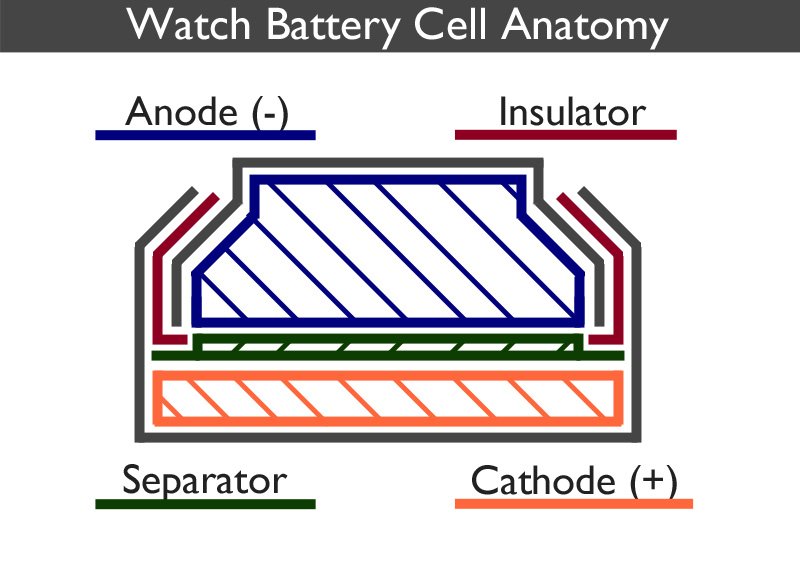 The watch battery is split into two distinct parts, the anode can (negative) and the cathode can (positive). The anode is a gel containing a mixture of the negative electrode (Silver Oxide) and the electrolyte (Sodium Hydroxide). The cathode contains the positive electrode (Zinc). Each part is surrounded by a metal shell, coloured grey in the above image, which forms the outside of the cell as well as acting as the corresponding positive and negative contact points.
The watch battery is split into two distinct parts, the anode can (negative) and the cathode can (positive). The anode is a gel containing a mixture of the negative electrode (Silver Oxide) and the electrolyte (Sodium Hydroxide). The cathode contains the positive electrode (Zinc). Each part is surrounded by a metal shell, coloured grey in the above image, which forms the outside of the cell as well as acting as the corresponding positive and negative contact points.
A separator, which is usually a mesh, prevents the electrodes from mixing but allows ions to flow between each half of the cell.
Although the electrodes do not touch one another, they are electrically connected by the electrolyte.
The insulator ensures that the two parts of the casing are held in place and do not touch one another. It also acts as a seal preventing the chemical mixtures from escaping. It is usually made from nylon. Sometimes it is referred to as the gasket.
As a battery cell ages the separator can degrade, this can allow some of the electrolyte in the anode to come into direct contact with the zinc in the cathode. When this happens electrolysis occurs.
Because electrolysis only occurs once the separator has degraded, the possibility of a battery leaking increases as the battery approaches its shelf life expiry. Other factors such as temperature and remaining capacity also contribute to the likelihood of the electrolyte escaping.
Electrolysis is a chemical reaction where the sodium hydroxide electrolyte splits and passes its oxygen onto the zinc.
In the below formula Na is Sodium, O is Oxygen, H is Hydrogen and Zn is Zinc.
NaOH + Zn = Na + ZnO + H
Sodium Hydroxide plus Zinc becomes Sodium plus Zinc Oxide plus Hydrogen
Hydrogen is the least dense element, in fact once separated from the electrolyte it takes up over 20 times more volume inside the battery case. As the cell is sealed the hydrogen cannot escape which causes a build up of pressure.
This pressure, as it tries to equalise itself with the outside air, will try and push its way out. As we can see from the diagram above, the insulator (coloured red) is the weak point, and this is where the gas will try and escape through. If the build up is significant enough, the insulator will rupture allowing the hydrogen gas to escape.
Once this seal is broken, the highly alkaline and corrosive electrolyte is then free to leak out of the battery. Any additional Hydrogen gas being produced will help to expel the electrolyte at a faster rate.

Mercury Content in Silver Oxide Cells

Watch Battery Electrolytes
There are two types of electrolyte used in Silver Oxide watch batteries. We have already discussed Sodium Hydroxide, which is the most common type, and is in use in the general low drain batteries for standard quartz watches.
Sodium Hydroxide can also be known as either Caustic Soda or Lye
In what are termed high drain batteries, Potassium Hydroxide (KOH) is used.
Potassium Hydroxide is sometimes called Caustic Potash or Potash Lye
The function of the electrolytes is almost identical in both low and high drain batteries, however in a low drain cell the internal resistance is higher, which limits the amount of energy that can be drained at any one time. High drain cells have a much lower internal resistance, which means that if a watch demands a large amount of energy, such as for sounding an alarm, then the battery will provide it.
Both electrolytes have a similar white crystalline appearance, have a comparable corrosive nature and both are used in the form of a slightly liquid gel which helps aid their conductivity. The fact that it is a liquid is what allows the electrolyte to flow and therefore leak.
In high drain batteries the separator is thinner, which allows the cell to provide larger pulses of energy if required. The downside to this is that it is more susceptible to degrade.
As a result: high drain batteries are significantly more likely to leak than low drain cells.

Safety Notice: care should be taken when touching a leaked battery, or any chemical residue, as both types of electrolyte will decompose proteins and lipids in the skin. These chemicals have a strong exothermic reaction as they dissolve in water; the heat from which can cause a chemical burn. Latex gloves or finger cots are therefore recommended when handling expended batteries. If your skin does come into direct contact with any leak from a battery you should dry brush any trace of chemical from your skin before you attempt to wash the area with water
Coin Cell Watch Batteries (Lithium)

Identifying Watch Battery Leaks







In this example the battery has also just started to leak, however you will note that in this case the negative battery contact has some tarnishing on it. As the battery contacts are an integrated part of the circuit board if this is not just some grease or dirt that can be cleaned off, but rather corrosion caused by a battery leak; then the entire circuit board needs to be replaced and this watch will need servicing



How to Clean a Watch Battery Leak



Watch Battery Button Cell Survey Results
A combined survey was undertaken to assess the issue of watch battery leaks. The outcome of the survey comes from the evaluation of over 3000 watches where the battery had expired. The results derive from a mix of sources from both North America and Europe including independent watch repair centres, official branded workshops and solo enterprises. The variety of watches tested covers the full-range of prices and brands that occupy the majority of the market segments.
In these results the use of the phrases such as “Best” or “Better than” relates only to the cell’s relative potential to leak, and should not be taken as a recommendation of which type or brand of cell to use. When selecting a battery the overall characteristics must be considered. Quality indicating factors such as the relative flatness of the discharge rate over the cell’s lifespan, or the total capacity of the battery, were not examined.
The conclusions will be presented as a comparison to the parts considered. This is because although there was some variance in the results based mainly on geographical area, the results were all in agreement when viewed in the form of a general list as exhibited below.
Many thanks to those individuals and businesses that participated in this survey.

As discussed earlier, the need for the separator to be thinner in a high drain battery is one of the major contributory factors that lead this battery type to leak more frequently than a low drain cell.

The height of the cell appears to be the main contributory factor in the likelihood of a battery leaking.
Examples; Wide and Tall 394/SR936SW, Wide and Short 371/SR920SW

Taken in isolation these results can be misleading. However when you note that most quartz Chronograph watches use Wide and Tall cells and quartz Gents movements generally use Wide and Short battery sizes ,the relationship between the type of movement and the chance of a leak occurring is more apparent

Only brands where more than 100 examples of drained batteries were examined are included. “Origin” was noted due to the clustering of results that became evident. This could be due to the regional exchange of technology and resources where the Button cells are manufactured. The difference in each of the “Rating” is based on the derived rate for the likelihood of a battery leaking, and are separated by a difference in the potential leaking rate of at least 3%.
Although not included in the results, unbranded batteries if grouped together were by far the most likely type to leak.


How to Reduce the Risk of your Watch Battery Leaking
You will never be able to completely exclude the risk of a Silver Oxide battery leaking. The following tips however can be followed to reduce the risk of the watch needing a full-service or exchange of movement:
- Do not leave a battery in a watch unless you intend to use it
- Do not wait until a battery has died before replacing it
- When fitting a battery, if possible use one that is less than 6 months old
- Any stocks of watch batteries should be refreshed every year
- Always use a branded battery
Always remember to recycle your old Silver Oxide button cells – even leaked ones. Not only does it stop harmful chemicals contaminating the environment, but the cells retain some value and so can be sold for scrap to an appropriate recycling company.






Helen Le Feuvre
11 June, 2014 at 5:45 pm
I purchased and expensive Omega watch and didn’t wear it. Two years later when I wanted to wear it had stopped. I took it in to have a new battery fitted, the shop sent it back to Omega who now say I need replacement works as the battery has leaked. Charge £320!!!
I had no idea this could happen until today. Buyers should be warned about possible leaky batteries.
I once had watch I bought on the beach in Thailand for £3 it lasted for ten years. I only replaced the battery once.
Which was the best buy?
Chris Morrow
26 September, 2014 at 2:34 am
I have a Seiko watch with has taken up stopping when I fly long haul routes in Asia. This is the second episode it has done it and the battery this time can be seen to leak. Sometimes returning to the ground it will start again but once it starts stopping you need a new battery. Has anyone else seen this?
Colin
28 September, 2014 at 8:55 pm
Hi Chris,
Thanks for your comment. If your watch is experiencing a change during a long flight, then it’s likely caused by the change of air pressure. This means two things:
Firstly, your watch is not sealed properly. Most watches should have some degree of water resistancy, but this ability relies upon rubber seals which degrade and perish over time.
Secondly, your watch battery was already leaking before your flight. The change in air pressure on a normal aeroplane will not be anywhere near significant enough to cause a battery to leak. However it will accelerate any leak that is already occuring.
So my advice would be to get your watch resealed, change the battery and have any leak cleaned; and remember to change your watch battery after 2-3 years.
You don’t wait for your car to breakdown before you get it serviced afterall 🙂
I hope that helps
Colin
Luigi
21 January, 2016 at 3:30 pm
Thank you very much indeed, Colin, for such a thorough, well written and illustrated piece on this matter.
If I may, I’d like to ask following image 9 starting from top: if small green specs on the negative testing point can be observed, will Rodico remove them or is it clear indicator that corrosion is underway along the entire negative contact underneath the blue circuit board and thus needs to be entirely replaced? In other words, once corrosion has begun on metal parts, what can be done?
Thank you in advance for your kind reply.
Luigi
Colin
22 January, 2016 at 12:04 am
Hi Luigi,
Thank you for your comment. Once corrosion has started there is not a great deal that can be done. It is not always an immediately fatal fault though, and so the watch can still work even after a battery has leaked and corrosion has started. If the leak is cleaned up properly, with peg wood and Rodico, it can stem the further increase of damage, however it is not something that can be repaired; and so as you mentioned, the circuit plus any other affected parts would need replacing when the watch eventually fails. Colin
Global Imports, Inc.
13 December, 2016 at 7:49 am
Many thanks for sharing helpful information about watch batteries, I am looking to read more post from you.
Anders Flint
4 February, 2017 at 1:19 pm
Interesting read, I have mostly mechanical watches but do have 2 basic quartz watches that my dad “forgot” to return when he left the armed forces. They both use eta 955.102 movements, I’ve always used Renata batteries (since they’re made by the same folks that make the movt) but I think now I’ll be switching to energiser to minimise the risk of leaks.
Colin
7 February, 2017 at 10:05 am
Hi Anders,
Although Swatch own both Renata and ETA, they both operate very independently from one another and so there aren’t any quality links between the two companies.
Renata is the industry standard, and almost every major watch brand uses them, although I’ve been suprised since publishing the article how many other watchmakers have told me that they always had a suspicion about the quality of Renata batteries. I’ve no interest in promoting one brand over another, but Energiser does seem to be a more stable choice. The 955.102 movement is now obsolete and so if you’re wanting to keep the original movement in the watch for as long as possible, then choosing a good battery will go a long way.
Colin
naidine
17 December, 2017 at 12:55 pm
Thanks for sharing your information, I really like the article, I got here while I was looking for answer to my concern. I have one of the Lionel railroad watches with tin case that were sold trough program ” As seen on TV”. It’s been many years that it is sealed in the box. I was looking to find out what type of battery the Lionel watch use and could battery leak while sealed in the box. Thanks I appreciate your help
Colin
7 January, 2018 at 10:28 am
Hi, I don’t know the watch brand, but I would take a guess that a watch sold through a TV channel would have been sourced from China. The movement inside is likely Japanese, and the battery would likely be a low drain silver oxide. So there is some risk of it leaking. There would be little reason for this type of watch to use a lithium battery.
The cause of a silver oxide battery leaking is internal to the battery itself and so it does not matter whether the watch is sealed or not, however it is has been kept somewhere cool then that may have helped.
A battery leaking is not a certainty though, but if you do not intend to use the watch I would recommend getting the dead battery removed from the watch as a precaution.
Colin
Elizabeth Ferrigno
2 April, 2019 at 10:05 am
I have a Cartier watch that stopped. I took it to my jewellers where I always go and was told the battery had leaked and damaged some of the movement so needed a clean, service and new parts at a cost of £325. I really think we should be made aware that watch batteries can leak. I had no idea this could happen.
Colin
3 April, 2019 at 9:43 am
Hi Elizabeth,
Thanks for your message. Yes it’s a shame. Some batteries are more prone than others but most watch batteries will leak and can damage the watch beyond repair at some point. For brands, such as Cartier, they would feel that it is not in their interest to inform you about a possible flaw with their watch; plus they would want to encourage the customer to get their watch regularly serviced with them or to even purchase a new watch if they damage is sufficient enough.
Colin
David Snell
14 May, 2019 at 6:05 pm
Greetings! Last weekend Sunday I bought a really cheap and old not working Casio CA-901 that is in need of a lot of TLC. This evening I removed both batteries and found both had leaked. On contacts, I found area where batteries had leaked had formed crystals already. I then took a cotton swab and cleaned away acid crystals. Area is now clean as a whistle and does not look like any damage to circuit or battery contacts. Do you think there is a possibility that battery leaks did not spread deeper than onto contacts?
Colin
14 May, 2019 at 6:24 pm
Hi David,
Thanks for your comment. Well done on cleaning the area up, you may well be lucky. My article is speaking mostly from a commercial situation and how a high-end watch should be treated; to always err on the side of caution and assume that a battery leak is almost always significant and the watch will require servicing.
The case may be that you have got lucky and that the damage on the contacts; which will have spread, has at least not caused damage that is significant enough to stop the watches from working. Corrosion on the contacts will at a minimum cause the resistance of the circuit to increase which will also increase battery consumption. A lot of the older Casio models are now obsolete and so they would be difficult to find parts to repair them if the damage was more significant.
The only way for you to be sure is to put a new battery in and keep your fingers and toes all cross.
Best of luck!
Colin
David Snell
14 May, 2019 at 6:41 pm
Hello Colin. You are most welcome! Thank you for your reply also. I collect and repair vintage digitals as best I can as a hobby. No commercials on my side! haha! You hit the nail right on the head. Vintage Casio is a near to impossible task in finding parts. Only options I have are either to steal parts from a less valuable digital or to improvise (I made an alarm coil not too long ago using spring from a watch bar of the same size). Ebay is a wondeful place to find “new old stock” on some parts. Sadly not all. To err on the side of caution is best and I will let you know if battery replacement remedied my vintage purchase. Thank you again for your reply.
albone UK
3 July, 2019 at 7:50 am
I really like your site and have book marked it. There are a lot of learning here.
Abraham
15 August, 2019 at 11:58 pm
I’ve had really expensiveSwiss watches.
And cheaper Seiko/citizen as well as really cheap casios. All Swiss stopped working, needed either a battery or movement changed. The Seiko/citizen needed just batteries every 2 years, and the casios are still running. Years later n still ticking.
Makes you wonder.
Lloyd Robinson
14 September, 2019 at 8:10 pm
My battery leaked in a Tissot watch. I have not cleaned it. Only replaced the battery . It is only when I did further research that I realised that damaged has been done to my watch mechanisms. The second hand is no longer moving correctly. Do I need to replace the mechanisms or have a service?
Colin
16 September, 2019 at 8:58 am
Hi Lloyd,
Thanks for your message. Once a watch battery has leaked, then unless it is cleaned the contacts and eventually the circuit will continue to be damaged. Unfortunately once it reaches the stage where it is affecting the function of your watch then it is usually too late, and all the damaged parts will need to be replaced.
Generally speaking when a battery powered watch is serviced then the entire movement is replaced each time; it is simply more economical to do this than take the time to dismantle a watch, identify the problem, and repair the movement.
The best place to get Tissot watches repaired is the main Tissot Service Centre based in Southampton.
I hope that helps.
Colin
Rene Bondoc
9 January, 2020 at 2:52 pm
If the battery terminal is broken in a watch can this be repaired?
Rene Bondoc
Colin
10 January, 2020 at 2:33 pm
Hi Rene,
Generally not. In most cases the battery terminal is part of the circuit of the movement, and so to replace the terminal you must replace the entire circuit. In such cases where the movement or parts are no longer available for replacement, then you could solder the terminal to repair it, however this will only be a temporary repair as this weak point will likely break again in the future.
Colin
Nancy
18 September, 2020 at 1:20 am
Question for Colin… I took my RADO watch in for a battery replacement because it was starting to run slow and it had been about 2years so the battery needed to be replaced. It was working when I gave it to a very reputable jeweler in the Detroit area. i was to pick it up in an hour as they were to replace the battery in house while i waited. They called me and told me they tried 3 new batteries and it would not work. Needless to say i was shocked it was working when i dropped it off… They said they would send it out for an estimate to have it repaired. it took 2 weeks to get the estimate and they called me and said that the battery had leaked and caused a lot of damage and needed an over haul at the cost of $256. My question is can a watch continue to work if the battery leaked this bad to need an overhaul? Could the jeweler have done something to cause this when they opened it and tried to replace the battery in house? i have seen many posts about not letting a dead battery sit in a watch as it will eventually leak, but can a watch work if the battery is leaking?
Please let me know what you think of the situation. Your comment would be greatly appreciated.
Colin
21 September, 2020 at 11:54 am
Hi Nancy,
Thanks for your question.
A battery in a watch will generally last for up to 2 years from new; there can be exceptions, lithium batteries last longer, and watches without a seconds hand tend to last a little longer too, and some watches can last for 5 years on the same battery – although this is unusual.
A battery powered watch doesn’t run slowly unless there is an issue with it. You mention that you took it to a reputable jeweller, which is a good sign. You’ve said that it is working when you dropped it off, but the fact that it was running slowly – for a number of years – shows that it wasn’t really working properly. A battery that has leaked can corrode the watch, that means that when you remove the battery then part of the contacts in the watch can come away too. That means that as soon as you remove a leaked battery, it will not work it you place a new battery in there.
Leaking batteries can still work, so that doesn’t prove or disprove anything. The jeweller could have damaged the watch when the battery was being changed, but all things considered I would think that they are telling the truth and your watch needs a service. Watches are very delicate instruments, and it doesn’t take much to upset them or stop them from working.
I hope that helps.
Colin
mark j. dombek
26 October, 2021 at 12:04 am
Colin,
Thank you for this very well written and illustrated article. Since I began to work on watches in 2003, hundreds of battery changes have also been done. All along it has been common to find leaking off brand batteries as well as quite a number from Renata, Rayovac and Varta. It has been rare to find a leaking Energizer, Sony, Seiko and Maxell. If so, it was when the watch had sat a very long time when discharged and allowed to get quite warm, as when kept in a car’s glovebox or left setting in sunlight on a window ledge.
Because of the Swiss reputation for guality, initially, a considerable stock of Renata batteries was puchased. After a couple of years of having watches coming back with leaking batteries, the switch was made to Energizer and no problems since.
Recently I decided to give Maxell a try, but this was before reading your article and seeing the comparative leakage chart. Now, I wish I had stayed with Energizer.
Thanks again for the great article,
M.J. Dombek (Milwaukee, Wis.)
Colin
9 November, 2021 at 11:12 am
Great, thanks Mark, I’m glad you found the article useful.
Colin
Kevin
2 December, 2022 at 3:55 pm
Hello Colin-
I had a Victorinox Peak II watch with erratic starting and stopping (with newish battery) over a 2 year period. It also had the inside crystal slightly fogged due to some “moisture(?)” intrusion. A nearby jeweler changed battery and cleaned inside of crystal and said, it was difficult to clean. An authorized repair facility later deemed the movement as unsalvagable and had me replace it. The watch internals never seemed to show water intrusion, but I wonder if battery gassing off (hydrogen?) could cause slight (but noticeable) fogging on inside of crystal?
This discussion underscores that quality quartz movement maintenance should not be taken for granted or ignored.
It consultancy Australia
3 January, 2023 at 5:06 am
I am so glad you shared this information. It is very helpful for me.
Gary Ronning
4 January, 2024 at 10:15 pm
Hi Colin – I am enjoying your commentaries. I have the opportunity to acquire 4 Zenith quartz watches very reasonably, which evidently have had battery leakage. These are from my watch repair jeweler. Assuming the worst, what might the charges be for repairing/replacing the movements? The watches are otherwise in good condition. I also have an older (no date) Longines Conquest watch with the same issue. Any info would be appreciated. Thank you ! Gary (USA)
Colin
24 January, 2024 at 5:08 pm
Hi Gary, the cost of the movements can vary wildly depending on the current availability. Some Zenith watches use ETA Quartz movements, such as a 955 type or similar. These are only around $20-30 each, and so quite affordable to replace, although if you have an unusual type it may be necessary to send the watch to the manufacturer for them to replace/repair the watch. Colin
Selvia Chapman
20 January, 2026 at 6:52 am
I gave my Dad a Timex Expedition for his anniversary quite a few years ago. He loved it because the watchface numbers glowed in the dark eventually it stopped running. When he passed away this past May2025. It was given back to me as part of my inheritance. I noticed it had something green on the band does this mean that the batteries leaked? If so if cleaned up could it run again with a new battery?I found the same watch new on eBay would it be possible to use the new parts to repair the old one. I just want it to run again it’s very special to me.